HTML5 Canvas Physics animation
When the name below is clicked, the animation made by 'HTML5 Canvas' is displayed.
Please click "Return" of a browser when it returns to this
menu panel.
|
 |
HTML5 Canvas |
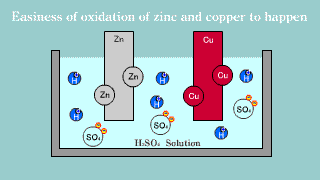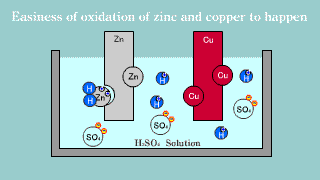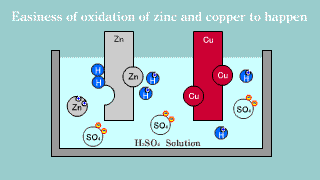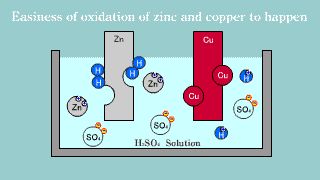
| Because zinc Zn puts out the electron, it becomes an ion, it melts, and
the electron is gotten, it becomes a hydrogen gas, it extracts, and copper
is not changed into the hydrogen ion, it is zinc that the electron is lost
and is oxidized easily. |
|
|
 |
|
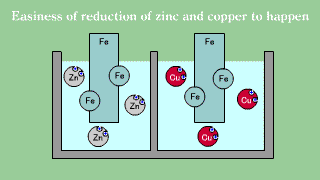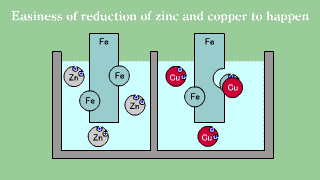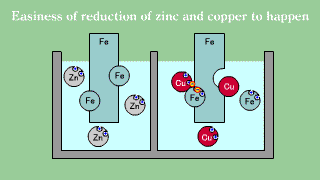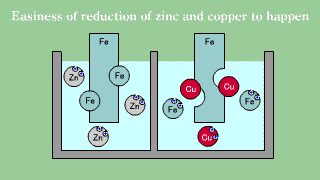
| Iron is put out the electron, becomes an ion, and melts, and when iron
Fe and copper (2) ions Cu2+ exists together, the electron is gotten and
extracted by copper (2) ion though doesn't change even if iron Fe and zinc
ion Zn2+ exists together it. |
|
|
 |
|
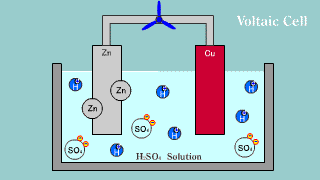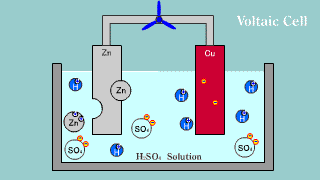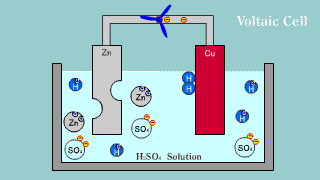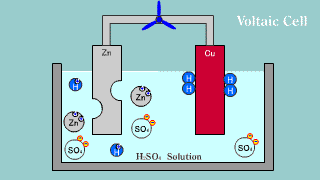
| The Volta battery is put out the electron by zinc Zn, becomes an ion,
and melts, and the hydrogen ion is gotten the electron, becomes a hydrogen
gas, and is extracted. |
|
|
 |
|
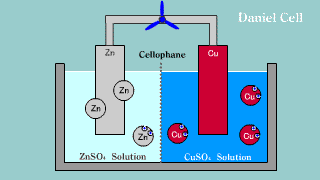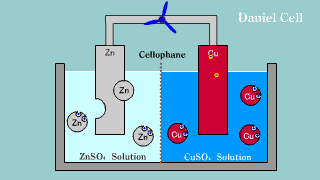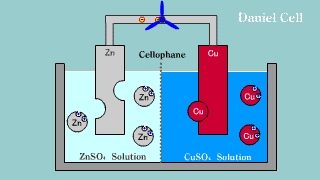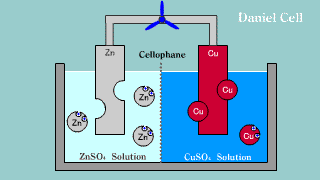
| Daniell cell is put out the electron by the difference of the ionization
potential of zinc Zn and copper Cu by zinc, becomes an ion, and melts,
and the copper ion gets and extracts the electron. |
|
|
 |
|
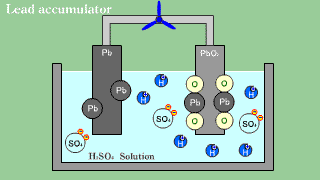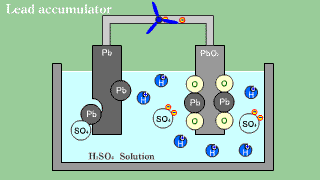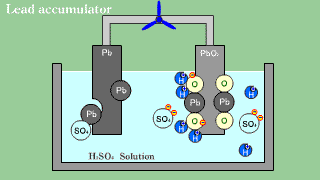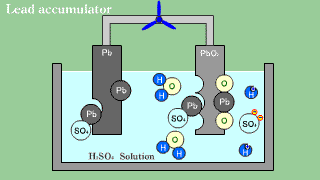
| As for the lead accumulator, both lead Pb becomes an anode in the cathode
lead oxide PbO2, and becomes sulfuric acid lead PbSO4 by the electrical
discharge. |
|
|
 |
|
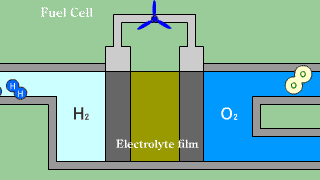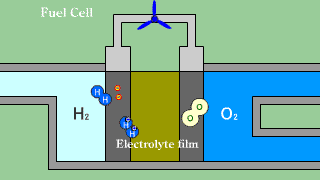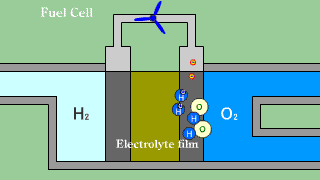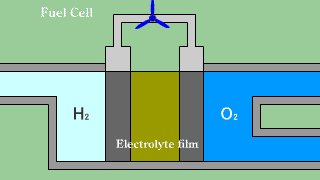
| When putting out the electron when hydrogen H2 that is the fuel passes
the electrolyte film, uniting with oxygen O2, and becoming water H2O, the
electron is received. |
|
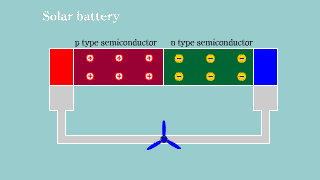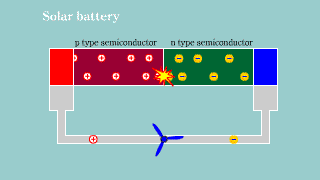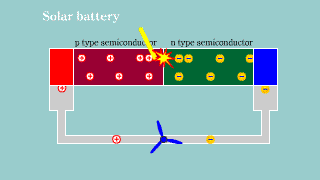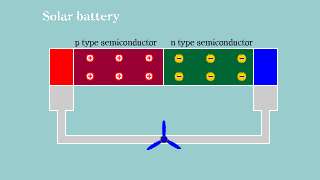
 Solar battery Solar battery |
 |
|
| If light hits the semiconductor to which pn is connected, the hall and
the electron appear, and it becomes a battery. |
|
 Electroanalysis of water Electroanalysis of water |
 |
|
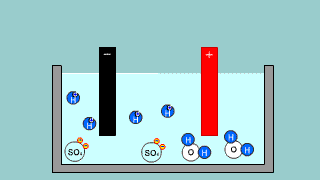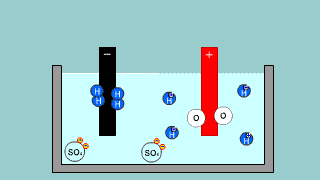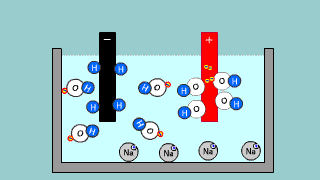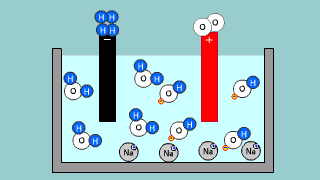
| It is an explanation of electroanalysis of water with sulfuric acid H2SO4
and electroanalysis of water by sodium hydroxide NaOH. |
|
|
Electromagnetism Battery Wave Sound wave Light Wave Mechanics Thermology Special relativity To top page
|







